 
When one atom connects with another atom, they make up a chemical element. What Are Atoms and What is Their Significance?Īn atom is one of the most significant things in the world as it is the smallest unit of matter. We first help students identify the basic parts and then work on how electron configuration affects the chemical nature of substances. We will also look at how this affects an atoms location on the Periodic Table of Elements. We will advance on to looking further into the nucleus and explore nuclear chemistry of atoms that are not very stable. Students will learn how to display valence shells with Lewis Dot diagrams. We will also look at how electrons are positioned and concept of orbital diagrams and how to determine valence shell configurations and what this means about the nature of the atom. We will look at how mass number affects the balance of the atom. Students will learn how to predict the element formed based on the number of protons an atom has. We break down the anatomy of these structures to display this for students and we will explore the Bohr model of this structure. We look at the function of each of the subatomic particles and how they interact to form molecules and ions. 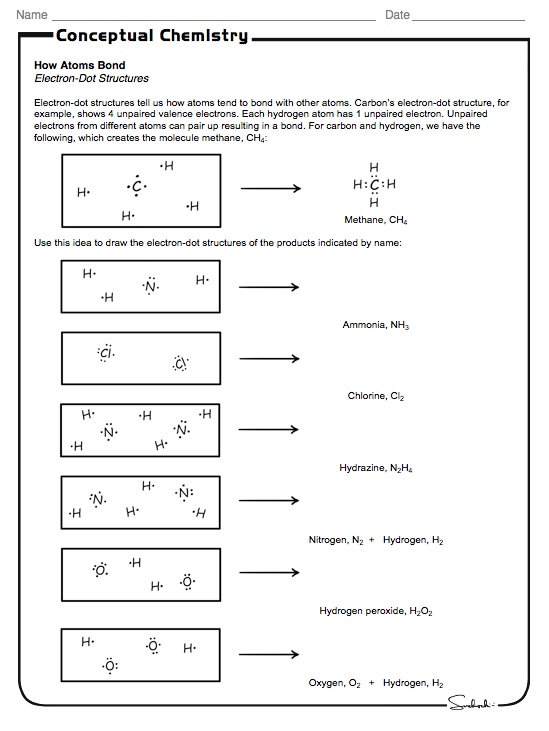
These worksheets have students explore the nature of atoms and their structure. (ii).This is truly one of the largest collections of atomic structure worksheets in one place. Thus, atom consists of a heavy positively charged nucleus in the centre containing all protons and the electrons were revolving around the nucleus so that the centrifugal force balances the force of attraction.Ītomic number and mass number: The general notation that is used to represent the mass number and atomic number of a given atoms is $_\] It was therefore, concluded that there was sufficient empty space within the atom and small heavy positively charged body at the center called nucleus. 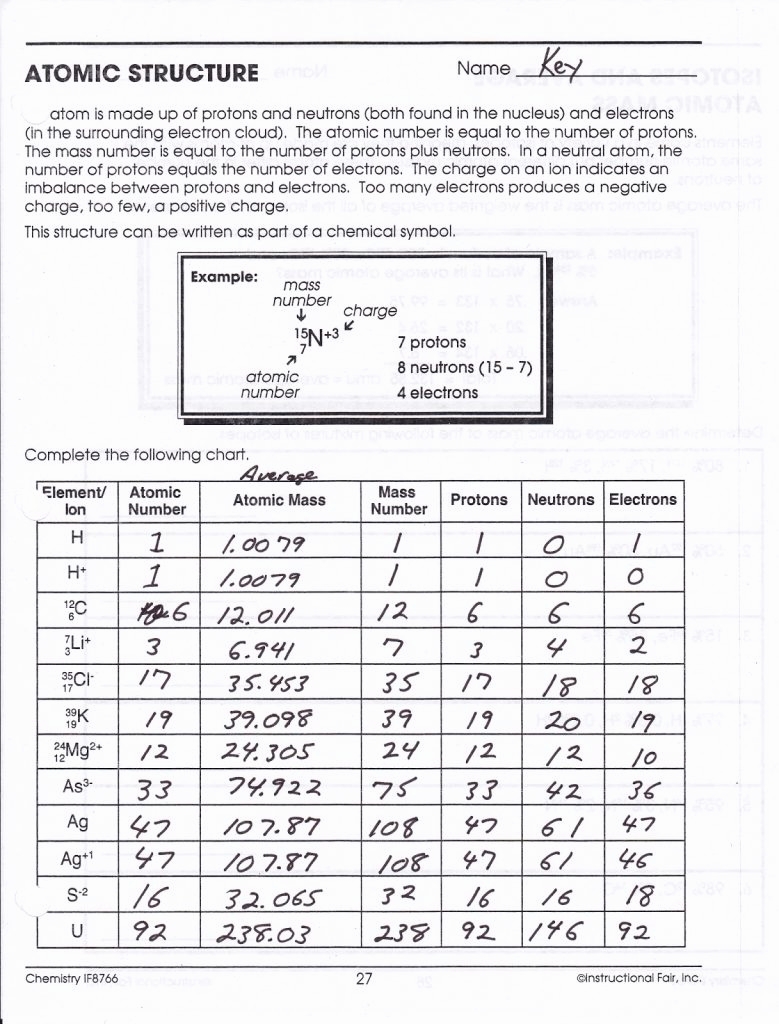
Most of the particles passed through the foil undeflected, a few were deflected through small angle while very few were deflected back. Rutherford’s model of atom: A thin foil of gold was bombarded with -particles. Rutherford’s -rays scattering experiments. Thomson’s model of atom: An atom is a sphere of positive electricity in which sufficient number of electrons were embedded to neutralize the positive charge just as seeds in a melon or raisins in pudding. Neutron: A fundamental particle which carries no charge but has a mass nearly equal to that of hydrogen atom. Proton: A fundamental particle which carries one unit positive charge and has a mass nearly equal to that of hydrogen atom. It is made up of electrons, protons and neutrons called fundamental particles.Įlectron: A fundamental particle which carries one unit negative charge and has a mass nearly equal to 1/1837th of that of hydrogen atom. Structure of Atom Chapter at a Glance - Class 11 NCERT SolutionsĬonstituents of atom: Atom is no longer considered as indivisible. You can also download the Structure of Atom Class 11 questions and answers PDF, and go through them. Our subject matter experts will get the right answers for all the questions that are asked in the examinations relating to Class 11 Chemistry Chapter 2. The Class 11 Chemistry NCERT Solutions Chapter 2 are prepared by the experts of Vedantu who are working in this field for decades now. NCERT Solutions for Class 11 Chemistry Chapter 2 are available for you to download online in PDF format. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
